Why does radius increase with higher atomic numbers in a group? As you move down a group in the periodic table, additional layers of electrons are being added, which naturally causes the ionic radius to increase as you move down the periodic table. While the atomic radius follows a similar trend, ions may be larger or smaller than neutral atoms. 
(b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. But for the nonmetallic elements, the ionic radius increases because there are more electrons than protons. Figure 2.8.2: Definitions of the Atomic Radius. More protons are added, but the outer valence shell remains the same, so the positively charged nucleus draws in the electrons more tightly. Moving down a group one finds that like-charged ions grow in size, as expected by their larger orbitals. 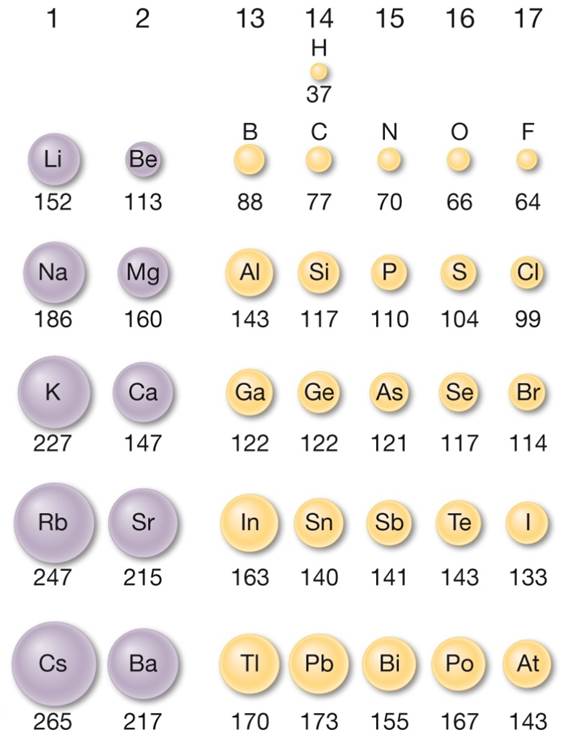
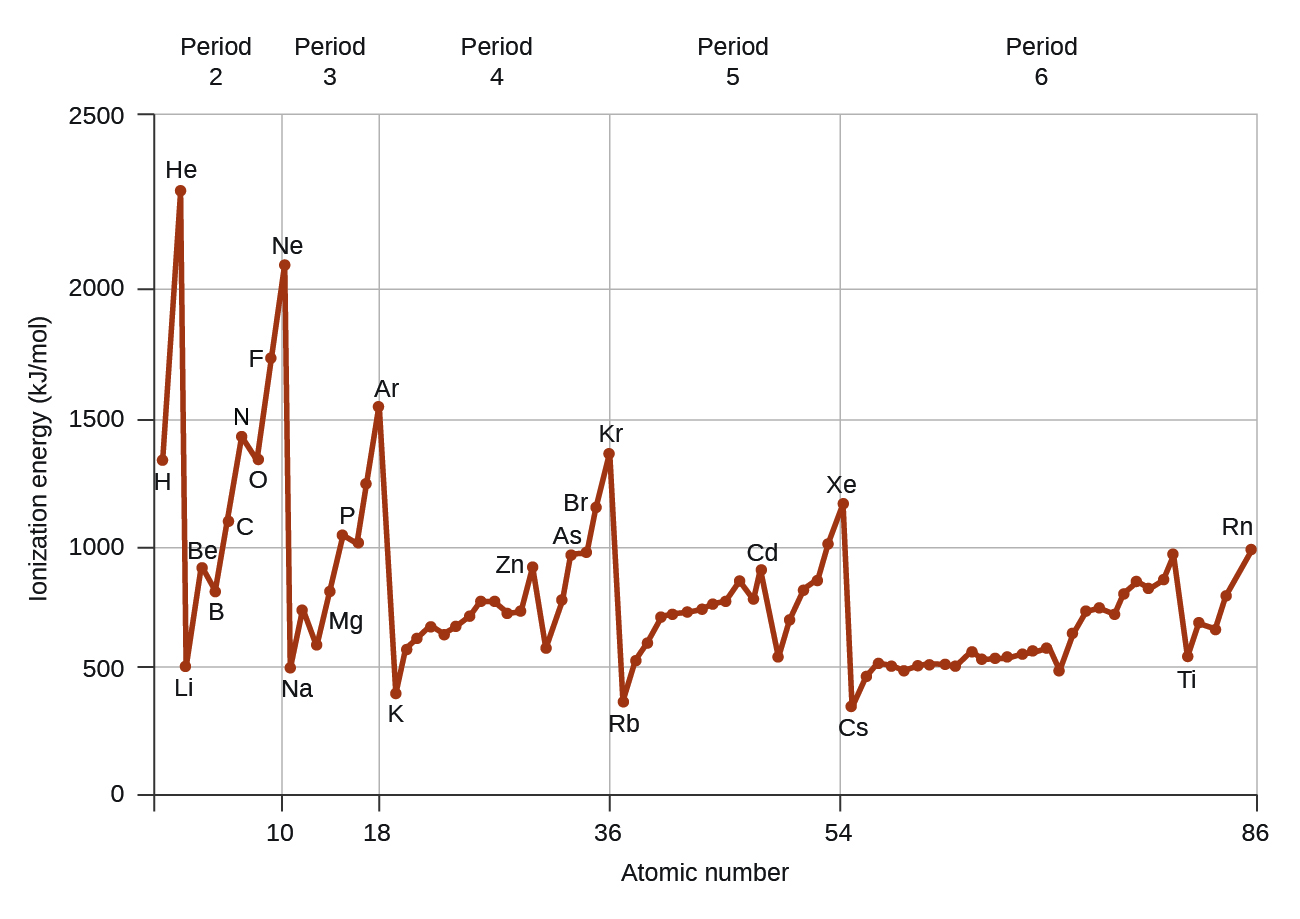
E r > E r + and E r E r-To a limited extent, trends in ionic sizes follow those of atomic sizes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
